 |
Name: | Wei Zhou | |
| eMail: | wei.zhou@catalysis.de | ||
| Institution: | Leibniz Institute for Catalysis Rostock (LIKAT) | ||
| Start Date: | 1/7/2016 | Return | |
| Supervisors: | Dr. Henrik Junge | ||
| Prof. Dr. Matthias Beller | |||
Base metal-catalysed hydrogenation and dehydrogenation of liquid hydrogen carriers
|
Hydrogenation and dehydrogenation reaction belong to the most important reactions with respect to the synthesis of bulk chemicals. Recently the application with respect to hydrogen storage in liquid hydrogen carriers earned more attention. Based on the expertise in homogeneous hydrogenation and dehydrogenation reactions the activities in the group of Henrik Junge will focus onto the development of active and stable catalysts for the hydrogenation of CO2/bicarbonate to liquid hydrogen carriers like formic acid or methanol and the dehydrogenation of the resulting products back to hydrogen and the respective byproducts, e.g. CO2. This will allow these reactions to proceed under ambient conditions and make them applicable for energy storage devices.
So far mainly noble metal containing catalysts have been applied in these reactions, but also iron, nickel or cobalt complexes could constitute appropriate catalysts for these reactions. The focus will lie on the substitution of noble metals, which is important. With respect to the availability as well as for economic reasons the catalyst synthesis for these reactions will be based on transition metals like Fe, Co, Ni, Mn and Mo. For the hydrogenation of CO2/bicarbonate and the dehydrogenation of HCO2H/formate especially tripodal and multidentate ligands, like triphosphines, tetraphosphines, aminopyridines, tetramines will be applied. For the dehydrogenation of MeOH the focus will lie e.g. on pincer type ligands. The catalyst development will be accompanied by kinetic and mechanistic studies to increase the activities and stabilities based on the knowledge how the catalysts act. Thus the catalyst systems can be improved in an iterative process.
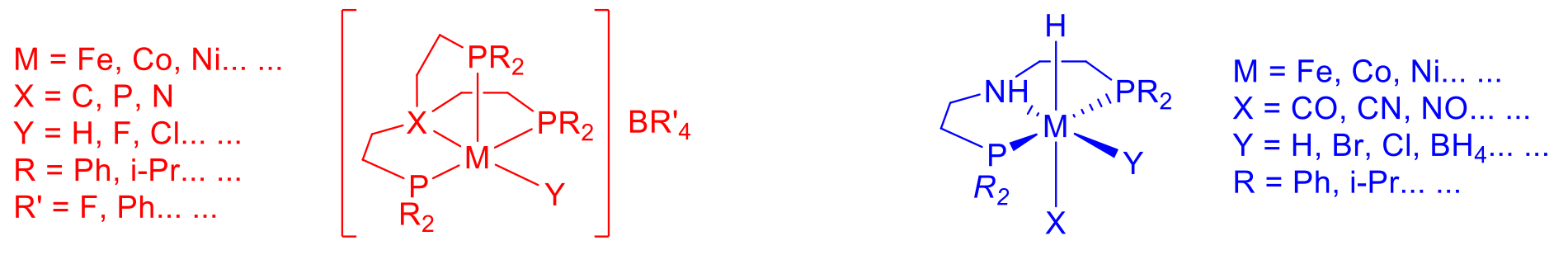 |
| Catalysts designed for (de)hydrogenation reactions |